
- Service
Research Programs
We pursue high-impact scientific initiatives to advance solutions for patients living with the effects of PTSD and TBI.
Brain trauma affects millions. Learn about traumatic brain injury (TBI) and post-traumatic stress disorder (PTSD) and how they impact civilians and Veterans.

Understand brain trauma (TBI/PTSD) and the current state of research.
TBI symptoms, diagnosis, treatment, and the impact of our research.
PTSD symptoms, diagnosis, treatment, and the impact of our research.
Learn about suicide risk among military Veterans with brain trauma.
A new Community Coordination model to accelerate a first generation of diagnostics and treatments for Traumatic Brain Injury.
Cohen Veterans Bioscience is a non-profit 501(c)(3) biomedical research and technology organization dedicated to advancing brain health by fast-tracking precision diagnostics and tailored therapeutics.

Explore our Mission & Vision to advance solutions for brain trauma.
Meet the minds behind CVB.
Meet our Board of Directors.
A voice for the Veterans community.
Read stories from people living with brain trauma.
Interested in joining the team? Explore careers at CVB.
Connect with the experts at CVB.
Our approach is to build enabling platforms with strategic partners and to adopt a team science approach to fast-track solutions in years, not decades.

Fast-tracking diagnostics & therapeutics for brain trauma.
Advocating at the federal, state, and local levels.
Helping high-impact research succeed.
Driving quality & reproducibility in science.
Explore our research publications.
Advancing our understanding of invisible wounds.
Learn how to participate in clinical trials.
Fostering a collaborative approach to research.
Donate today to advance solutions for brain trauma. Together, we can advance research and improve lives.

Get the latest updates in PTSD & TBI research.
Share your story: how has TBI impacted you?
We look forward to hearing from you.
Learn about the latest news & events from CVB.
View press releases and more.
A new Community Coordination model to accelerate a first generation of diagnostics and treatments for Traumatic Brain Injury.
Cohen Veterans Bioscience is doing things differently. Our approach is to build enabling platforms with strategic partners and to adopt a team science approach to fast-track solutions in years, not decades.
We bring together the brightest minds and deploy the most innovative scientific tools with an emphasis on scale, risk-minimization, efficiency and speed to advance our understanding of PTSD and TBI. Through rigorous research and breaking down traditional silos, we’re working to bring more accurate diagnostic tests for PTSD and TBI to patients using big data, digital health and innovative technology to match patients to the right treatment.
Moving away from the one patient, one treatment approach.
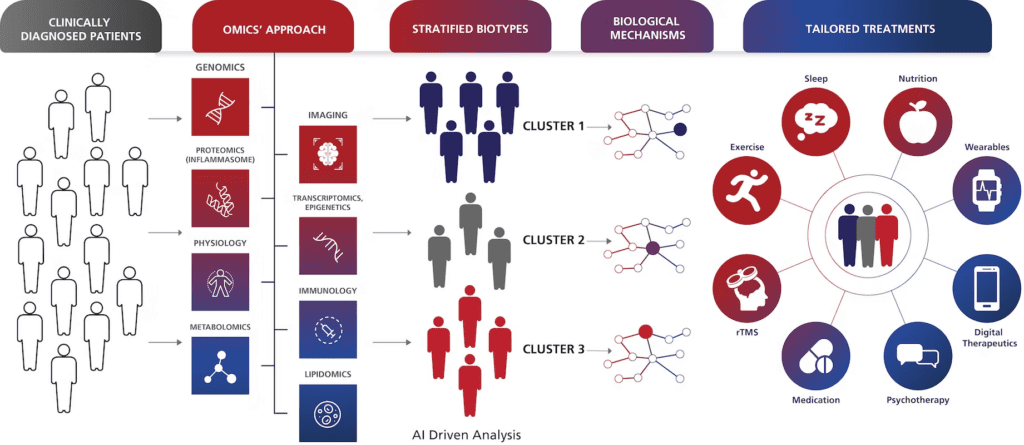
Our research and advocacy aim to improve the lives of all people who experience the effects of trauma by:



Our research to fast-track diagnostics and treatments for PTSD and TBI can be applied to advance solutions for other brain disorders.

Peter Hoehn
Chief Executive Officer
Peter Hoehn
Chief Executive Officer
Peter Hoehn is a proven healthcare business leader with 25+ years’ experience in a variety of commercial and strategy roles, predominantly at Johnson & Johnson. Throughout his career Peter has demonstrated the ability to develop insight driven strategies in complex markets and mobilize internal and external partners to achieve results. He brings broad-based commercial, strategy and leadership experiences across Pharmaceuticals, Diagnostics, Digital Health and Precision Medicine, including brand positioning and messaging, reimbursement and market access, global launches, organizational capability building, business model innovation, new opportunity evaluation, business development, and policy.
Peter started his career as a corporate lawyer and entered the pharmaceutical industry as an attorney for Bristol Myers Squibb. After a short time as a lawyer Peter moved into various strategy, marketing and sales management roles at BMS before joining Johnson & Johnson in 2003. He started at J&J as Global Commercial Leader for Reminyl, an Alzheimer’s drug, and was then promoted to Global Marketing Leader, Neurology, where he had global commercial responsibility for in-line neurology products as well as products in late stage of development.
In 2008 Peter joined J&J’s Ortho Clinical Diagnostics organization as Vice President for WW Marketing and Strategic Marketing, responsible for long-term organizational strategy and a large global diagnostics business. In 2012 Peter moved back to J&J’s pharmaceuticals business as Precision Medicine Commercial Strategy Leader. In this role, Peter helped develop J&J’s Precision Medicine strategy, and built and led the Precision Medicine Commercial Team which was responsible for leading strategic marketing and market access for companion and complementary diagnostics across all Therapeutic Areas.
In his most previous role before joining CVB, Peter was Head, Commercial Strategy for J&J Science for Minds, a new organization focused on innovative development models and digital solutions to address Serious Mental Illness and dementia. In this position he developed the strategy, portfolio recommendations, investment approach, external collaborations, and business models for new mental health and brain health digital business ventures.
Peter received a BA in Government and Economics from the College of William and Mary, and a JD from New York University.

Nicole Harmon, PhD
President
Nicole Harmon, PhD
President
Nicole Harmon brings more than 18 years of experience in psychology, philanthropy, patient advocacy, public health, and clinical research, most recently as the Chief Operating Officer and Chief of Staff for Clinical Data Interchange Standards Consortium (CDISC), FDA’s partner for data standards. During her tenure there she transformed CDISC’s financial roadmaps, program management and global brand. Additionally, she developed and managed complex partnerships with leading U.S and International funding partners, U.S. FDA, Japan PMDA, European Medicines Agency, NIH, WHO, EU’s Innovative Medicines Initiatives, Global Patient Foundations and others to drive data standards and sharing initiatives and oversaw grants cultivation and management successfully achieving funding for and administering multiple large-scale projects. Prior to her time with CDISC, Dr. Harmon evaluated patients with Traumatic Brain Injury and Psychiatric conditions in a post-acute rehabilitation hospital. Nicole earned her doctorate in Counselor Education and Supervision at Texas Tech University with dissertation focus on understanding the caregivers experience associated with acquired brain injury following discharge from rehabilitation.

Magali Haas, MD, PhD
Founder & Senior Advisor
Magali Haas, MD, PhD
Founder & Senior Advisor
Dr. Magali Haas is Founder and Board Chair and former CEO of Cohen Veterans Bioscience, a non-profit biomedical and technology research organization based in New York City whose mission is to fast-track diagnostics and therapeutics to advance precision brain health with a focus on the “invisible wounds” of post-traumatic stress and traumatic brain injuries. She currently leads the National TBI Action Alliance and serves as a Senior Strategy & Science Advisor to the CEO. Dr. Haas is also Founder and Scientific Lead of the Brain Health Nexus, a global initiative to galvanize science and data collaboration, to optimize brain health across the life course and prevent and treat brain diseases and disorders, through the establishment of a brain health index. Dr. Haas has over 15 years of pharmaceutical executive experience, predominantly at Johnson & Johnson, where she assumed broad end-to-end leadership roles in early and late-stage neuroscience clinical development, translational medicine, diagnostics and integrative solutions. She serves on several international advisory boards including The European Platform for Neurodegenerative Diseases (EPND), VirtualBrainCloud, Krembil Centre for Neuroinformatics (based in Canada), and IMEC for nanoelectronics. She is a frequent speaker and invited participant at conferences and symposia around the world. Dr. Haas earned her BS in bioengineering from the University of Pennsylvania, an MS in biomedical engineering from Rutgers University, and her MD and PhD in neuroscience with distinction from Albert Einstein College of Medicine.

Allyson Gage, PhD
Chief Medical Officer
Allyson Gage, PhD
Chief Medical Officer
Dr. Allyson T. Gage is a neuroscientist and drug development executive, who has over 18 years of experience leading teams in all phases of clinical development. She has been responsible for the overall strategic and clinical development of small molecules, biologics, and cellular therapies for the treatment of central nervous system disorders, including depression, alcohol dependence, Alzheimer’s dementia, neuropathic pain, traumatic brain injury, and spinal cord injury. Allyson has a proven track record in partnering with therapeutic area clinicians, US and International regulators, patient groups, and data scientists to translate pre-clinical information into human evaluation, to design informative clinical trial protocols, and ensure a regulatory path with approvable and clinically meaningful outcome measures. Prior to joining Cohen Veterans Bioscience, Allyson’s most recent experience was focused in the field of regenerative medicine, working toward the development of a therapeutic for spinal cord injury. Allyson earned her BA from Rutgers College in New Brunswick, NJ and her MS and PhD in neuroscience from Albert Einstein College of Medicine, New York.

Lee Lancashire, PhD
Chief Information Officer
Lee Lancashire, PhD
Chief Information Officer
Lee Lancashire, PhD has over 15 years of experience in machine learning and statistics. His PhD was focused on the development of artificial neural network based methodologies where he was involved in many of the first applications of neural nets to high dimensional datasets in bioinformatics. Prior to joining Cohen Veterans Bioscience, Lee spent over five years within the IP&Science business of Thomson Reuters where he established and led the machine learning and statistical data analysis team. Here he held global responsibility for managing and delivering analytical projects, serving the majority of the top 10 pharmaceutical companies, as well as government, academic and non-profit organizations. These projects focused on utilizing advanced machine learning approaches in the molecular profiling of disease for biomarker discovery, patient stratification and drug target identification. Lee completed his post-doctoral research in Biostatistics and Bioinformatics at the Paterson Institute for Cancer Research, a Cancer Research UK core facility. He has also worked as a bioinformatics project lead in personalized medicine and diagnostics companies CompanDX and Almac Diagnostics. He has published in over 30 key industry journals and holds several patents around the use of neural networks in the biomedical domain. In his free time Lee devotes time volunteering as a trustee for the Ichthyosis Support Group, a charity that supports individuals and families affected by a group of rare skin disorders.

Eugene Rakhmatulin
Chief Technology Officer
Eugene Rakhmatulin
Chief Technology Officer
Eugene Rakhmatulin is launching a new department at Cohen Veterans Bioscience dedicated to highly sophisticated technology development. He has more than two decades of experience in Information Technologies (IT), including 15+ years in Life Sciences field. Eugene’s work has been largely focused on software development, systems architecture and management. Previously, Eugene was the Senior Director of Enabling Technologies and Informatics Delivery at Clarivate Analytics, formerly Thomson Reuters Intellectual Property & Science business. In this role, he led a global Professional Services Technology organization that was dedicated to designing and implementing data integration solutions, analytics tools and content services for major pharmaceutical companies, government, academia and non-profit organizations. Eugene joined Thomson Reuters in 2010 through an acquisition of GeneGo, the leading provider of software and content for Systems Biology. There, he served as the Head of IT and software Development. Eugene earned his MS in Computer Science and Engineering from the Moscow Aviation Institute (State Technical University) in Moscow, Russia.

Mark Zervas, PhD
Head of Discovery & Preclinical Research
Mark Zervas, PhD
Head of Discovery & Preclinical Research
Mark Zervas earned his Ph.D. in Neuroscience at the Albert Einstein College of Medicine, was a postdoctoral fellow at the NYU School of Medicine, and was the Manning Assistant Professor of Biology in the MCB department at Brown University. Mark served as Senior Scientist at Amgen, where he led a research team that used human genetics to discover novel therapeutic targets and human iPSC-derived dopamine neurons for understanding target biology and mechanisms underlying Parkinson’s Disease, including mitochondrial dysfunction. Mark was then a Senior Scientist at Alnylam Pharmaceuticals where he led a team using RNAi to treat ocular diseases. At CVB, Mark is responsible for identifying putative therapeutic targets and advancing biological understanding of PTSD and TBI using multiple modalities.

Ragini Verma, PhD
Head of Biomarkers & Clinical Development
Ragini Verma, PhD
Head of Biomarkers & Clinical Development
Ragini Verma, PhD, brings her expertise in neuroimaging, biomarkers, preclinical and clinical analytics and clinical translation. She has a double masters in math and computer applications, a PhD in computer vision and two postdoctoral fellowships, one in video-based computer vision, and the second in neuroimaging. Prior to joining CVB, Ragini was a tenured professor of Radiology and Neurosurgery at the University of Pennsylvania (and is now an adjunct professor). At Penn, she led a team developing tools and conducting multimodal image analytics to create precision imaging solutions for clinical needs, and for biomarker discovery in disorders like autism, brain cancer and traumatic brain injury. She also led a translational center, that was tasked with deploying advanced AI-based solutions translating whole body medical imaging to real world clinical practice with the goal of empowering physicians and improving patient outcomes. As an adjunct professor, she will maintain her brain cancer, autism and TBI neuroimaging marker discovery portfolios to complement her work at CVB.

Nicole Bjorklund, PhD
Director, Translational Research & Development
Nicole Bjorklund, PhD
Director, Translational Research & Development
Nicole Bjorklund, PhD, brings her expertise in biochemistry, translational neuroscience and biomarker development to the RAPID-Dx program, focused on advancing biomarker discovery and validation in post-traumatic stress disorder and traumatic brain injury. Prior to joining CVB, Nicole led the development and management of the Diagnostics Accelerator initiative at the Alzheimer’s Drug Discovery Foundation, which supports the advancement of biomarkers for use in drug development for Alzheimer’s disease and related dementias. Nicole started her career as an Assistant Research Professor and Operations Director of the Biomarker and Biorepository core facilities at Albert Einstein College of Medicine. Her laboratories, as part of the CTSA-supported Institute of Clinical and Translational Research, supported the large number of clinical research studies at Einstein and Montefiore Medical Center in the Bronx, NY. Nicole completed postdoctoral training at the University of Texas Medical Branch, where she investigated molecular resistance mechanisms to Alzheimer’s disease. She earned a bachelor’s degree in chemistry from Boise State University and a doctorate in biochemistry at Washington State University.

Sanjiv Bhave, PhD
Senior Program Manager, Translational Research & Development
Sanjiv Bhave, PhD
Senior Program Manager, Translational Research & Development
Sanjiv Bhave, Ph.D. is an experienced neuroscientist and all throughout his professional journey, across different continents, he has worked, in academia and industry, in a wide range of fields in neuroscience, molecular biology, biochemistry, genomics and proteomics. His background, a unique blend of disciplines such as neurosciences, genetical genomics, transcriptomics/proteomics and bioinformatics, enables him to quickly take the lead on any type of project – “traditional” hypothesis driven projects or “global” hypothesis driven projects such as those designed to assess “candidate genes or biomarkers”. He has worked at the University of Colorado School of Medicine for twenty plus years on a number of different aspects of neuroscience including developing a database, and associated data analyses tools, for the whole transcriptome in the whole brain of close to hundred different strains of mice and rats. He earned his Ph.D. in Biochemistry from the MS University of Baroda, India and did postdoctoral studies at the Downstate Medical Center, SUNY, Brooklyn and Wayne State School of Medicine Detroit.

Kevin Urban, PhD
Director, Data Science
Kevin Urban, PhD
Director, Data Science
Kevin began his career developing space weather prediction models using a diverse range of complex data sets stemming from NASA and NOAA spacecraft missions in Earth’s ionosphere, magnetosphere, and interplanetary space, as well as from large-scale networks of ground-based instrumentation. During this time, Kevin also published on space mission design and dynamical system modeling of granular fluid systems. After receiving his PhD in physics from New Jersey Institute of Technology and Rutgers University, Kevin worked as a data scientist at WWE, where he built predictive models for subscriber behavior on the WWE Network (a monthly, direct-to-consumer, video streaming service averaging ~2 million daily active subscribers) and led various data engineering efforts, such as the collection, ingestion, and harmonization of WWE fan data across a host of social media and online platforms. At Cohen Veterans Bioscience, Kevin has worked with wearable sensor datasets in effort to identify “digital biomarkers” that might be used to track health status and disease progression.

Andrew Thompson, PhD
Associate Director, Data Science
Andrew Thompson, PhD
Associate Director, Data Science
Andrew is a data scientist leading the RAPID-Dx program advance biomarker discovery and validation in post-traumatic stress disorder (PTSD) and traumatic brain injury (TBI). Previously, he worked at PatientsLikeMe where he led analyses of patient-reported health data and multi-omic biological data to define predictive biomarkers and characterize disease sub-types. During his graduate degree training in neurobiology, Andrew focused on plasticity in the developing visual system, investigating a role for feedback projections in the fine-tuning of circuits during development. Prior to his PhD, Andrew worked with the discovery group at Vanda Pharmaceuticals, where he performed pharmacogenomics research in support of a phase 3 clinical trial and expression profiling studies – all in an effort to elucidate common gene signatures and repurpose existing drugs for new indications. Andrew earned his BS in biology from the University of Maryland and his PhD in biological and biomedical sciences from Harvard University.

Emanuele Libertini, PhD
Associate Director, Data Science
Emanuele Libertini, PhD
Associate Director, Data Science
Emanuele has a background in bioinformatics, network biology, genomics, metagenomics and epigenomics. He has worked in the academic field at University College London and Institut Pasteur Paris before moving to the private sector with Thomson Reuters and Clarivate. His interests lie in integrating and interpreting biological datasets representing multiple levels of regulation of molecular mechanisms with quantitative methods suited to capture resolution-dependent signals. His experience includes exposure to modelling plant signalling networks, host-pathogen relationships in the infection of human cells, and epigenomic regulation in humans. In the last five years he has focused on modelling molecular mechanisms of disease in a broad range of pharma projects, including pathologies of the brain and the nervous system.

Ryan Wales, PhD
Data Engineer
Ryan Wales, PhD
Data Engineer
Ryan Wales, PhD has 10 years of experience in neuroscience research, including 4 years studying PTSD models in rodents and 6 years studying functional neuroimaging in humans. Prior to joining CVB, he used structural and functional MRI to study how the brain and cognition change with age and disease. Specifically, Ryan is interested in biomarkers of neurodegenerative diseases, including Parkinson’s disease and Alzheimer’s disease. His dissertation focused on the effects of Alzheimer’s-related proteins, amyloid-beta and tau, on functional network connectivity among individuals without dementia. Ryan earned his BS in Neuroscience and a minor in Psychology from the University of California, Riverside and his PhD in Cognitive Neuroscience from Stony Brook University.

Zhanna Rozenberg
Senior Director, Technology Development
Zhanna Rozenberg
Director, Technology Development
Zhanna received her BA at Cornell University where she triple majored in Economics, Comparative Literature, and an College Scholars major that tread on philosophy, critical theory, and sociology. She completed a minor in East Asian Studies. She later went to study statistics, econometrics, and machine learning at the Ruprecht-Karls-Universität Heidelberg, Universität Mannheim, and the Metis’s data science bootcamp. Zhanna is interested in making technology that helps people live more meaningful lives, using data to improve human performance. Zhanna is active in NY’s meetup culture focusing on data science, neuroscience, wearables, IoT, and habit design. Zhanna’s major individual project, “Hindsight”, explored the interaction between subjective and objective measures of human performance.

Matthew Springer
Senior Manager, Technology Operations
Matthew Springer
Senior Cloud Engineer, Technology Development
Matthew Springer has over 25 years of experience in the IT industry. He has extensive experience in computer manufacture, IT infrastructure management, database administration and cloud engineering. Prior to joining Cohen Veterans Bioscience, he was a Senior Development Operations Engineer in the Life Sciences division of Clarivate Analytics, formerly the Thomson Reuters Intellectual Property & Science business. He was responsible for the design and implementation of the cloud infrastructure within the Professional Services organization. He was also responsible for the management of their database infrastructure and the automation of their software deployments. Matthew joined Thomson Reuters in 2011 as a Senior Database Engineer. Matthew is involved in various California-based initiatives to feed and support the homeless community. He also has a keen interest in Cinematography, Videography and Photography.

Isabel Curro
Senior Manager, Technology Development
Isabel Curro
Senior Manager, Technology Development
Isabel is working with the Digital Health team on device validation, data analysis and protocol development for internal testing and Sleepwell projects. Previously, she completed an engineering internship with VERO Biotech, a medical device start-up based in Atlanta. Isabel also conducted research on how to combat antibiotic- resistant bacteria and how to develop nanoparticles for targeted drug delivery at the Lab for Synthetic Immunity at Georgia Tech. She also interned with Siemens in Erlangen, Germany. Isabel received a BS in Biomedical Engineering with a minor in International Affairs from the Georgia Institute of Technology where she participated in the honors program and international plan. In addition, Isabel studied abroad at UC3M in Madrid, Spain.

Maryan Zirkle, MD, MS, MA
Executive Director, BRAINCommons
Maryan Zirkle, MD, MS, MA
Executive Director, BRAINCommons
Maryan Zirkle MD, MS, MA is an accomplished professional with more than 10 years of experience working in the research industry. She has expertise in research network infrastructure development and sustainability, Big Data, patient-centered outcomes research, healthcare delivery and medicine, mental health, interdisciplinary-based team management, contract management, and active research portfolio management.
Before joining Cohen Veterans Bioscience, Dr. Zirkle was the Associate Director of the Research Infrastructure team at the Patient-Centered Outcomes Research Institute (PCORI). She was responsible for providing intellectual and organizational oversight for the development and regulation of PCORI’s National Patient-Centered Clinical Data Research Network, PCORnet. Zirkle has also worked as a research health science specialist at the Department of Veterans Affairs. She gained considerable experience in polytrauma, traumatic brain injury (TBI), posttraumatic stress disorder (PTSD) and medical informatics research at Department of Veterans Affairs (VA) medical centers in Tampa, Florida, and Portland, Oregon. She completed a three-year advanced fellowship in clinical informatics at the VA in Portland. She also received two bachelor of arts degrees from the University of Florida, a master of arts in rehabilitative and mental health counseling from the University of South Florida, a master of science in biomedical and clinical informatics from Oregon Health and Science University, and a medical degree from Spartan Health Sciences University.

Chantelle Ferland-Beckham, PhD
Senior Director, External Affairs
Chantelle Ferland-Beckham, PhD
Senior Director, External Affairs
Dr. Chantelle Ferland-Beckham is a translational neuroscientist by training, with over 15 years of experience in preclinical models of neuropsychiatric disorders and brain injury models. She is also an experienced public policy and advocacy professional, having spent nearly 10 years working on advocacy and public policy issues at the local, state and federal levels. After earning her PhD in Neuroscience from Tulane University, she went on to complete postdoctoral fellowships in both traumatic brain injury and clinical and preclinical models of comorbid addition and post-traumatic stress disorder (PTSD). She was later selected to be an AAAS Science & Technology Policy Fellow at the National Institutes of Health. In this role as a Health Science Policy Analyst, she evaluated research programs and developed policies to promote progress in biomedical research and ensure the effective use of taxpayer funds to conduct research for the benefit of human health. She also assisted with Congressionally mandated reporting and liaison activities to strengthen the relationship between the NIH and Congress. Additionally, she worked on the Fellows in Innovation initiative, spearheaded by the Obama administration’s Office of Science and Technology Policy, to analyze current strategies and provide policy recommendations for suicide prevention in current and former members of the US military. In 2017, she joined Cohen Veterans Bioscience (CVB), serving in a capacity that bridged her scientific and public policy backgrounds and supporting both the programmatic and policy initiatives of CVB.

Lauren J. Goldsmith
Senior Director, Contracts and Grants Management
Lauren J. Goldsmith
Senior Director, Contracts and Grants Management
Lauren is a contracting professional with over 18 years in the pharmaceutical industry with specialized expertise in outsourcing and contracting for all R&D activities for clinical and preclinical trials. She also has experience in supporting corporate agreements, research collaborations with academic institutions, and vendor relationship management.
Before joining Cohen Veterans Bioscience, she was the Global Head of Site Contracts leading an international department of over 25 people globally.
Lauren graduated from Harvard University with a Bachelor of Liberal Arts and holds a Paralegal Certificate from Northeastern University. She has also holds a Certificate in the Fundamentals of the Bioscience Industry from The Center for Biotechnology from Stony Brook University.

David Gordon
Operations Manager
David Gordon
Operations Manager
David Gordon was born and raised in Orange County, California. In 2017, he came to New York City to attend university for politics and public policy. Beginning in 2018, David has worked in various political and electoral roles including director of field organizing, campaign manager and policy analyst. He has also worked as an aide to the top staff of foundations, community organizations and consultancies in both California and New York. He is a passionate advocate for affordable housing in New York City, and yet he maintains his California roots as a diehard fan of the Los Angeles Dodgers.

Michael Carson
Head of Communications
Michael Carson
Head of Communications
Michael Carson is the Head of Communications of Cohen Veterans Bioscience, the New York-based non-profit organization dedicated to advancing brain health.
Prior to Cohen Veterans Bioscience (CVB), Michael served as chief marketing officer of the Alzheimer’s Association leading strategic initiatives that drove the global conversation of early detection, diagnosis of Alzheimer’s disease and other dementia, and awareness initiatives in support of brain health. Michael’s role supported organizational advancements in advocacy and research.
Michael led the Association’s organizational branding, global communications and digital revenue streams. In addition to publishing initiatives Michael oversaw alz.org, social platforms, global media relations, celebrity engagement, advertising and internal communications for all staff.
Under Michael’s leadership, the Association has grown concern about Alzheimer’s disease and awareness of the organization and its programs and services. Reach among the public has significantly increased and Michael made significant social media gains in reach, engagement and revenue growth. Michael’s forward-thinking approach to new programs and initiatives has heightened concern, reduced stigma and advanced the cause.
Prior to joining the Alzheimer’s Association, Michael led marketing and operations as executive vice president and chief marketing officer for an ad- supported tech-based start-up venture. He also held marketing leadership roles at FTD.com, Ogilvy & Mather, Orbitz Travel and FCB.
He served on the Google App Developer Council, the Discover Financial Services Merchant Advisory Board and the Meta Social Good roundtable.
Michael received a master’s degree in business administration from the University of Notre Dame and a bachelor’s degree in business administration and marketing from Marquette University.

Elinor Litwack, CPA
Finance Comptroller (Contract)
Elinor Litwack, CPA
Finance Comptroller (Contract)
Elinor Litwack is Partner at GRF CPAs and Advisors’ outsourced accounting and advisory team. Over the past decade she has specialized in providing financial and organizational solutions to nonprofit organizations, utilizing state of the art technology combined with deep industry expertise. She is proficient in systems design, cloud computing, audit readiness and accounting best practices. As contract finance manager, Elinor leads the finance team at CVB.
Elinor serves as Treasurer of I Support the Girls, a nonprofit organization that facilitates donations of bras and menstrual hygiene products to women and girls experiencing homelessness. A graduate of George Washington University, Elinor was nominated for the Greater Washington Society of CPAs’ Women to Watch Awards in 2015. Additionally, Elinor was recognized in NYN Media’s 2021 Nonprofit 40 under 40.
Elinor is a member of the American Institute of Public Accountants, the Maryland Association of Certified Public Accountants and the New York State Society of CPAs, where she serves on the Chief Financial Officer committee. Additionally, Elinor participated on the planning committee for the 2021 Better Business Bureau of New York’s Charity Effectiveness Symposium and is serving as the 2021 Digital CPA Advisory Board Chair. Elinor lives in Englewood, New Jersey with her husband and four kids.





















Sorry, we have no current job openings. Thanks for your interest and please check this page for updates.
Cohen Veterans Bioscience is a non-profit 501(c)(3) biomedical research and technology organization dedicated to advancing brain health by fast-tracking precision diagnostics and tailored therapeutics.
These brave men and women voluntarily stepped up on the line to serve this great nation with the goal of protecting our freedoms…many have gone into harm’s way at personal expense and sacrifice. We cannot leave any more of them behind. It is our national obligation to support those that have come home with both visible and invisible wounds.
Frank Larkin
Member, Veterans Advisory Council



Jane Horton
Member, Veterans Advisory Council
Jane Horton
Member, Veterans Advisory Council
Ms. Jane Horton is a respected and influential global leader for national security, humanitarian, and veterans. A fearless and relentless advocate, she successfully fosters bipartisan collaboration on legislation, policies, and programs to better the lives of veterans, service members, their families, and those across the globe. Jane currently serves as a senior policy advisor in the United States Congress in Washington D.C. Previously, Jane was a senior advisor in the Office of the Secretary of Defense in the Pentagon. In this capacity, she advised on civilian and military personnel policy, military family policy, and international security affairs. She championed efforts to advance warfighter brain health and Gold Star family initiatives and has inspired or authored over twenty pieces of legislation in over a decade. Some of Jane’s additional experiences include serving as a special assistant to the Chairman of the Joint Chiefs of Staff, General Joseph Dunford, an advisor to the Chief of Staff of the Army, and the congressional and military liaison to the Ambassador of Afghanistan to the United States. She currently serves on the board of directors of numerous non-profits and is a proud member of Georgetown University's US- Afghan Women's Council.

Robin King
Member, Veterans Advisory Council
Robin King
Member, Veterans Advisory Council
Ms. King has been with the Navy SEAL Foundation since its inception in July 2000, and in that time has served as Chief Financial Officer, Deputy Executive Director, and Interim Executive Director before being appointed as Chief Executive Officer in May of 2013. Her exemplary leadership and commitment to financial transparency and accountability have allowed the Foundation to grow from a small organization focused on scholarships and educational opportunities to a multi-faceted programmatic foundation with dozens of specialized programs and an annual program budget in the millions.
Ms. King executes the daily mission of the Navy SEAL Foundation from Coronado, California, home of Naval Special Warfare Command (WARCOM), SEAL Qualification Training and the headquarters of NSW. She leads a staff of nearly twenty employees located throughout the United States. Under her command, the Navy SEAL Foundation has consistently achieved and maintained the highest rating possible from Charity Navigator, a national charity watchdog organization, placing it in the top 1% of all charities rated.
In August of 2019, Ms. King was awarded the prestigious United States Special Operations Command (USSOCOM) Outstanding Civilian Service Medal which recognized her steadfast dedication and superior service to the NSW community including raising millions of dollars to fund programs touching the lives
of tens of thousands of NSW warriors and families.
In 2017, Ms. King was awarded the USSOCOM Care Coalition Patriot Award. This distinguished award recognizes individuals and organizations who greatly support Special Operations Force (SOF) wounded warriors, their families, and surviving family members by providing significant and enduring
contributions that benefit their care and recovery. She is the first nonprofit leader to receive this award.
Ms. King is a current member of the Board of Directors for The Honor Foundation and the C4 Foundation, both nonprofits serving the Naval Special Warfare community.
Before her employment at the Foundation, Robin worked at Pacific Trust Bank, The Walt Disney Company, and McDonnell Douglas Corporation (Boeing). She holds a Bachelor of Science degree in Business Administration, Finance, from California State University, Long Beach.

Frank Larkin
Member, Veterans Advisory Council
Frank Larkin
Member, Veterans Advisory Council
Frank Larkin was formerly the United States Senate Sergeant at Arms (SAA). As chief law enforcement and executive officer of the Senate, the SAA enforces rules of the Senate; provides a range of technical and administrative services to Senators in their Washington, DC, and state offices; and maintains security in the Capitol and Senate office buildings.
Frank was a member of the Senior Executive Service, recently serving as both the Acting Director and the Vice Director of the Joint Improvised Explosive Device Defeat Organization (JIEDDO) within the Department of Defense. He previously served as JIEDDO’s Director for the Counter IED Operations-Intelligence Integration Center. JIEDDO’s mission was to focus all Department of Defense actions in support of the combatant commanders’ efforts to defeat improvised explosive devices (IEDs) as weapons of strategic influence. Frank’s focus was in support of our deployed warfighters and special operations forces around the globe and the integration of a “whole of government” disruption effort targeting extremist networks that employ IEDs.
Frank served for more than two decades in the United States Secret Service (USSS), beginning in 1984 as a Special Agent assigned to the Philadelphia and Washington Field Offices, before assignment to the Presidential Protective Division. He was assigned to the Office of Congressional Affairs for a two-year fellowship on Capitol Hill, serving a year each with the Senate and House Appropriations Committees. Frank entered the USSS supervisory ranks as Assistant to the Special Agent in Charge of the New York Field Office, where he led financial, electronic, and organized crimes investigations. Following the events of 9/11, he returned to Washington, DC, as the Assistant Special Agent in Charge of the Presidential Protective Division, supervising White House security operations. As Assistant Special Agent in Charge of the Technical Security Division, Frank was responsible for tactical countermeasures programs. In 2004, he was promoted to the Senior Executive Service, assuming the position of Deputy Assistant Director for Protective Research and Chief Technology Officer.
Following his USSS career, Frank was Director, Program Management & Leadership, for the Raytheon Company, and more recently, worked at Lockheed Martin’s Defense & Intelligence Solutions, responsible for providing operations and intelligence analysis support to the intelligence community.
A veteran of the US Navy, Frank has a significant military and law enforcement special operations background, serving as a special warfare operator in the Navy SEALs. After his Navy service, he was a uniformed patrol officer with the Norristown (PA) Police Department, a homicide detective with the Montgomery County (PA) District Attorney’s Office, and a Maryland State Trooper-Flight Paramedic.
Frank holds a BA degree in criminal justice and an MS degree in public administration from Villanova University. He is the recipient of numerous awards, including the US Secret Service award for valor, the Department of the Army’s Exceptional Civilian Service award, and the Superior Civilian Service award.

LTG John F. Mulholland, U.S. Army (Ret).
Member, Veterans Advisory Council
LTG John F. Mulholland, U.S. Army (Ret).
Member, Veterans Advisory Council
The proud son of a USAF fighter pilot and Korean War veteran, Lieutenant General Mulholland John Francis Mulholland, Jr. (Retired) was born in Clovis, New Mexico, but grew up in Bethesda, Maryland. He earned a Bachelor of Arts in History and was commissioned as a Second Lieutenant of Infantry, United States Army upon graduating from Furman University, Greenville, South Carolina, in 1978.
As a young Infantry officer, LTG Mulholland (Ret) served as platoon leader of both mechanized and airborne rifle platoons, as well as mortar platoon leader. Since joining the 1st Special Forces Regiment in 1983, LTG Mulholland (Ret) commanded from the captain through lieutenant general-level in Army and joint special operations forces, including serving as Deputy Commanding General, Joint Special Operations Command, as the Commanding General, United States Army Special Operations Command, and as the 15th Deputy Commander, United States Special Operations Command. Following graduating from the National War College in 2001, then-COL Mulholland assumed command of the 5th Special Forces Group, Airborne at Ft. Campbell, KY. In the immediate aftermath of the attacks of 9-11, Colonel Mulholland commanded Joint Special Operations Task Force-North (Task Force Dagger) in the opening days of Operation ENDURING FREEDOM and, later, Combined Joint Special Operations Task Force-West (Task Force Dagger) in the initial campaign of Operation IRAQI FREEDOM.
LTG Mulholland’s (Ret) overseas assignments included two tours in the former Panama Canal Zone, command of the 1st Battalion, 1st Special Forces Group (ABN), Okinawa, Japan and as the Chief, Office of Military Cooperation, Kuwait. LTG Mulholland (Ret) served at and completed his final tour on active duty as the Associate Director of Military Affairs, Central Intelligence Agency. LTG Mulholland (Ret) is married to the former Miriam Mitchell. Together, they have 4 grown children and 5 grandchildren. LTG Mulholland (Ret) and Miriam now operate Mulholland Consulting, LLC in Alexandria, Virginia.

Tim Szymanski, VADM, U.S. Navy (Ret.)
Member, Veterans Advisory Council
Tim Szymanski, VADM, U.S. Navy (Ret.)
Member, Veterans Advisory Council
Tim has served in many Navy and Joint Special Operations assignments as a Navy Special Warfare Officer (SEAL) for over 36 years. He most recently served as the Deputy Commander for United States Special Operations Command (USSOCOM) after serving as the Commander of Naval Special Warfare (NSW). In both roles he oversaw the manning, equipping and training, and employment of NSW and joint special operations forces, of 11,000 and 73,000 uniformed and civilian personnel, respectively.
He has an extensive background in raising, equipping, and training Navy, allied and joint special operations forces and has served operationally in Iraq, Afghanistan, Haiti, and other places of crises. As Deputy Commander USSOCOM and as Commander NSW he spearheaded force design and development initiatives, led organizational culture reviews and implementation plans, and developed leaders throughout the force.
Keenly aware of the detrimental effects of invisible wounds on Force and Family Readiness, he established policies and developed lines of effort to monitor, protect and enhance warfighter brain health.



Magali Haas, MD, PhD
Board Chair
Magali Haas, MD, PhD
Board Chair
Dr. Magali Haas is Founder and Board Chair and former CEO of Cohen Veterans Bioscience, a non-profit biomedical and technology research organization based in New York City whose mission is to fast-track diagnostics and therapeutics to advance precision brain health with a focus on the “invisible wounds” of post-traumatic stress and traumatic brain injuries. She currently leads the National TBI Action Alliance and serves as a Senior Strategy & Science Advisor to the CEO. Dr. Haas is also Founder and Scientific Lead of the Brain Health Nexus, a global initiative to galvanize science and data collaboration, to optimize brain health across the life course and prevent and treat brain diseases and disorders, through the establishment of a brain health index. Dr. Haas has over 15 years of pharmaceutical executive experience, predominantly at Johnson & Johnson, where she assumed broad end-to-end leadership roles in early and late-stage neuroscience clinical development, translational medicine, diagnostics and integrative solutions. She serves on several international advisory boards including The European Platform for Neurodegenerative Diseases (EPND), VirtualBrainCloud, Krembil Centre for Neuroinformatics (based in Canada), and IMEC for nanoelectronics. She is a frequent speaker and invited participant at conferences and symposia around the world. Dr. Haas earned her BS in bioengineering from the University of Pennsylvania, an MS in biomedical engineering from Rutgers University, and her MD and PhD in neuroscience with distinction from Albert Einstein College of Medicine.

Frank Larkin
Vice Chair
Frank Larkin
Vice Chair
Frank Larkin was formerly the United States Senate Sergeant at Arms (SAA). As chief law enforcement and executive officer of the Senate, the SAA enforces rules of the Senate; provides a range of technical and administrative services to Senators in their Washington, DC, and state offices; and maintains security in the Capitol and Senate office buildings.
Frank was a member of the Senior Executive Service, recently serving as both the Acting Director and the Vice Director of the Joint Improvised Explosive Device Defeat Organization (JIEDDO) within the Department of Defense. He previously served as JIEDDO’s Director for the Counter IED Operations-Intelligence Integration Center. JIEDDO’s mission was to focus all Department of Defense actions in support of the combatant commanders’ efforts to defeat improvised explosive devices (IEDs) as weapons of strategic influence. Frank’s focus was in support of our deployed warfighters and special operations forces around the globe and the integration of a “whole of government” disruption effort targeting extremist networks that employ IEDs.
Frank served for more than two decades in the United States Secret Service (USSS), beginning in 1984 as a Special Agent assigned to the Philadelphia and Washington Field Offices, before assignment to the Presidential Protective Division. He was assigned to the Office of Congressional Affairs for a two-year fellowship on Capitol Hill, serving a year each with the Senate and House Appropriations Committees. Frank entered the USSS supervisory ranks as Assistant to the Special Agent in Charge of the New York Field Office, where he led financial, electronic, and organized crimes investigations. Following the events of 9/11, he returned to Washington, DC, as the Assistant Special Agent in Charge of the Presidential Protective Division, supervising White House security operations. As Assistant Special Agent in Charge of the Technical Security Division, Frank was responsible for tactical countermeasures programs. In 2004, he was promoted to the Senior Executive Service, assuming the position of Deputy Assistant Director for Protective Research and Chief Technology Officer.
Following his USSS career, Frank was Director, Program Management & Leadership, for the Raytheon Company, and more recently, worked at Lockheed Martin’s Defense & Intelligence Solutions, responsible for providing operations and intelligence analysis support to the intelligence community.
A veteran of the US Navy, Frank has a significant military and law enforcement special operations background, serving as a special warfare operator in the Navy SEALs. After his Navy service, he was a uniformed patrol officer with the Norristown (PA) Police Department, a homicide detective with the Montgomery County (PA) District Attorney’s Office, and a Maryland State Trooper-Flight Paramedic.
Frank holds a BA degree in criminal justice and an MS degree in public administration from Villanova University. He is the recipient of numerous awards, including the US Secret Service award for valor, the Department of the Army’s Exceptional Civilian Service award, and the Superior Civilian Service award.

Michael Sullivan
Board Treasurer
Michael Sullivan
Board Treasurer
Michael C. Sullivan is the Chief of Staff and Head of External Affairs of Point72 Asset Management. Mr. Sullivan joined the Firm in 2007 after serving as a senior aide in the United States Senate, focusing on telecom, technology, and finance issues. Mr. Sullivan also served as a staff member in the U.S. House of Representatives working on similar issues. Before coming to Capitol Hill, Mr. Sullivan was the head of strategic development for a telecommunications focused trade association.
Mr. Sullivan is involved with several veterans’ mental health-related non-profit organizations. He helped establish and serves on the Board and as Treasurer of the Cohen Veterans Network, which operates a national network of clinics to treat veterans and their family members affected by Post-Traumatic Stress. Mr. Sullivan also serves as the Treasurer and on the Board of a second veterans-oriented non-profit, Cohen Veterans Bioscience, which is developing next-generation diagnostics and therapeutics to improve the detection and treatment of Post-Traumatic Stress.
Mr. Sullivan is an Associate at the NYU-Stern Endless Frontier Labs, where he mentors start-up companies in artificial intelligence, machine learning, and related fields. He is also a member of the Executive Committee of Students First New York, a nonprofit organization advocating for educational reform in New York State. Mr. Sullivan is a graduate of Vanderbilt University.

David M. Biondi, DO, FAAN
Board Member
David M. Biondi, DO, FAAN
Board Member
David Biondi received his medical degree from the University of New England, College of Osteopathic Medicine in Biddeford ME. He completed residency training in Neurology at the National Naval Medical Center in Bethesda MD and is board-certified in Neurology with subspecialty expertise in Headache and Pain Medicine. He received his Bachelor of Science – Magna Cum Laude in Pharmacy from the Long Island University, Schwartz College of Pharmacy & Health Science in Brooklyn NY.
Dr. Biondi has over 15 years of experience in clinical research and development of pharmaceuticals and medical devices in the neurology, psychiatry, and pain therapeutic areas. His experiences include the design, implementation, monitoring, and interpretation of phase 2 to phase 4 clinical trials and in-house and field-based activities associated with insights generation, new business opportunities, emerging health policy, knowledge translation and implementation, and organizational effectiveness. He previously served as Executive Director for Clinical Programs with Cohen Veterans Bioscience.
Dr. Biondi has over 20 years of clinical experience in medical practice and held medical directorships at the US Naval Hospital in Okinawa Japan, the Pain Management Program & Headache Institute at Maryview Medical Center in Portsmouth VA, the Pain Division of the Michigan Head Pain & Neurological Institute in Ann Arbor MI, and the Interdisciplinary Headache & Pain Rehabilitation Program at Spaulding Rehabilitation Hospital in Boston MA. He was previously an Assistant Neurologist at the Massachusetts General Hospital and an Instructor in Neurology at Harvard Medical School.
Dr. Biondi served as a trustee for the University of New England and was Chair of its Academic and Research Affairs committee and Vice-Chair of its Long-Range Planning committee. He also served as a Director and medical advisor for the After Action Network, a nonprofit Veteran Service Organization in Kansas City MO. He is a Fellow of the American Academy of Neurology.

Dave DeMarco, PhD
Board Member
Dave DeMarco, PhD
Board Member
Dr. Dave DeMarco is a senior advisor to C-suite leaders in the Life Sciences and Healthcare industries. He serves as Chief Strategy Officer for Material Health Strategies and works with c-suite executives on growth strategies and team performance in both the for-profit and non-for-profit industries. He previously served as Chair of the Board of the American Diabetes Association, led Ernst & Young’s (EY’s) Northeast Life Sciences practice and was the Deputy Leader of the US Healthcare Practice.
With over 25 years of leadership experience, Dave advises, coaches and shares insights on strategy and transformation, commercial operations, hHealthcare policy, governance, public-private-partnerships, and entrepreneurial growth.
While serving on the board of the American Diabetes Association, he led Board initiatives involving: CEO succession, Principal Officer succession, Board Development, Policy creation, strategic plan implementation and technology partnerships.
Dave has held key executive positions, including: Global Aesthetic Franchise Leader, Sanofi Aventis; US Health Deputy Leader, Northeast Region Life Sciences Practice Leader and Johnson & Johnson Global Client Services Partner, EY; Research Scientist, Internal Venture Leader, Amoco Chemical; and cofounder, Idea Connection Systems.
Dave worked closely with the Aventis NA leadership team to develop post-merger integration growth strategies and launch key blockbuster products aimed at, and succeeding in, doubling the size of the business in four years. As a senior partner in EY’s Global Life Sciences team, Dave piloted a new P&L accountable, go-to-market model, growing the Northeast practice at 30% CAGR, resulting in a $400 million business. In his service to Johnson & Johnson, he partnered with J&J leadership in developing Janssen Healthcare Innovation, charged with creating future business units. Dave led the transformation of EY’s US Health practice, resulting in a $750 million business. As a member of the Sanofi Aventis NA Dermatology leadership team, Dave developed the global aesthetic franchise strategy and served as the commercial head for Europe. For this turnaround project, he created a plan to triple EBITDA, implement new global pricing policies and double customer acquisition.
Dave received his B.S. from Grove City College and Ph.D. in chemistry from Purdue University.


By combining the power of biotechnology with rigorous research, high-performance computing, and data analytics, we’re accelerating the development of precision diagnostics and tailored treatments for military service members, Veterans, first responders, and all people living with the effects of brain trauma.
We advocate for people with brain trauma through a focused advocacy strategy that includes our Veterans Advisory Council, ongoing outreach to Congress, and dedicated educational programs.
Our goal is to advance tailored treatments for PTSD and TBI so that a clinician can target the right treatment to the right person at the time when they need help most.

CVB will achieve our mission to deliver solutions to people suffering from brain trauma by leading in multiple areas:

Researching and developing objective diagnostic tools that detect the underlying biology of brain trauma and treatments, including preventative wellness, digital health, drug and device approaches.
Convening the brightest minds in the PTSD and TBI research communities because collaboration, data sharing, and leveraging each partner’s unique capabilities is the fastest way to deliver impactful solutions to patients.


Developing research tools & platforms which leverage our own technology and innovative approaches to enable our partners to conduct research that can scale & accelerate the development of effective solutions for brain trauma.
Shaping the environment for cutting-edge research by providing education to researchers on best practices and by fostering support for advancing brain trauma solutions at the federal, state and local levels through advocacy and education.

Learn more about our research here. Or make a difference and support our work by making a donation today.
It seems we can't find what you're looking for.
New York Office (Main Office)
535 8th Ave, 12th floor
New York, NY 10018
Phone: (646) 970-4325
Cambridge Office
1 Broadway, 14th Floor
Cambridge, MA 02142
Cohen Veterans Bioscience (CVB) is a non-profit 501(c)(3) public charity research organization and does not offer medical advice. CVB encourages you to seek medical advice from a physician or healthcare provider if you have questions regarding a medical condition, or to call 911 or go to the nearest hospital if you find that you or someone you are concerned about is in an emergency situation.
Cohen Veterans Bioscience accepts requests for funding proposals by invitation only. CVB strategically leads and funds projects that align with our mission and current initiatives.
Cohen Veterans Bioscience is a non-profit 501(c)(3) biomedical research and technology organization dedicated to advancing brain health by fast-tracking precision diagnostics and tailored therapeutics.
Cohen Veterans Bioscience is a non-profit 501(c)(3) biomedical research and technology organization dedicated to advancing brain health by fast-tracking precision diagnostics and tailored therapeutics.
©2023 Cohen Veterans Bioscience | Privacy Policy | Terms of Use
